Looking at their structure, carboxylic acids are important organic compounds, and they are acyl derivatives. They are a kind of building materials for many derivatives, including esters and amides. In nature, we can find a whole range of various carboxylic acids, such as acetic acid CH3COOH, which is the basic compound of table vinegar, butyric acid CH3CH2CH2COOH responsible for the smell of rancid butter, or more complex cholic acid, which is the basic component of human bile juices.

The general formula of carboxylic acids is:
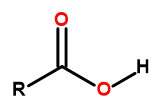
Figure1 The general formula of carboxylic acids is:
Nomenclature of carboxylic acids
According to the IUPAC nomenclature system, carboxylic acids can be named in two ways, depending on the size and complexity of the molecule. Acids that are aliphatic derivatives are named systematically by changing the alkane ending from -ane to -oic and adding the initial word “acid”, for example, propanoic acid, butanoic acid, 3-ethyl-6-methyloctanedioic acid. The numbering of carbon atoms in such chemical compounds always starts with the atom in the –COOH functional group. The second naming system, especially useful for acids in which the carboxyl group is attached to the ring, is permitted to add the words “acid” before and “carboxylic” after it to the systematic name of the compound. In such cases, the numbering of the carbon atoms begins with the atom to which the carboxyl group is attached, which itself is treated as a substituent.
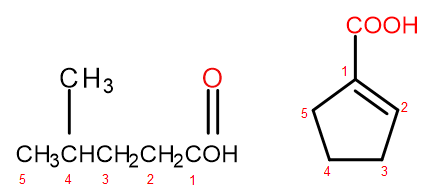
Figure 2 Numbering of carbon atoms in, from left to right: 4-methylpentanoic acid, 1-cyclopentenecarboxylic acid.
Due to the historical outline according to which carboxylic acids were among the first isolated, purified and characterized organic compounds, the IUPAC system also allows many common names for both organic compounds and their acyl groups.
Table 1 Selected examples of common names of carboxylic acids and their acyl groups.
| Carboxylic acid | Acyl group | ||
| Structure | Name | Structure | Name |
| HCOOH | Formic | HCO- | Formyl- |
| CH3COOH | Acetic | CH3CO- | Acetyl- |
| CH3CH2CH2COOH | Butyric | CH3CH2CH2CO- | Butyryl- |
| HOOOCCOOH | Oxalic | -OCCO- | Oxalil- |
| HOOC(CH2)2COOH | Succinic | -OC(CH2)2CO- | Succinyl- |
The structure and physical properties of carboxylic acids
The similarity of the carboxyl group to both ketones and alcohols may suggest similar physical properties. As in ketones, the carbon atom of the functional group has an sp2 hybridization. This is directly related to its flat structure and angles of about 120o between the C-C-O and O-C-O bonds. For example, acetic acid CH3COOH has bond angles equal to:
- 119o for C-C=O,
- 119o for C-C-OH,
- 122o for O=C-OH.
The bond lengths are as follows:
- 52Å for C-C,
- 25Å for C=O,
- 31Å for C-OH.
Carboxylic acids show their similarity to alcohols due to their strong associativity caused by the possibility of forming hydrogen bonds. In practice, most of these compounds exist as cyclic dimers whose structure is maintained by the formation of hydrogen bonds. Such a specific system of hydrogen bonds also affects the boiling points of carboxylic acids. It causes their significant increase compared to the corresponding alcohols. For example, physical constants – the melting point and the boiling point in degrees Celsius are respectively:
- Formic acid: 8.4; 100.7,
- Acetic acid: 16.6; 117.9,
- Propanoic acid: -20.8; 141,
- Benzoic acid: 122.1; 249.
Dissociation of carboxylic acids
The acidic properties of this group of compounds make it possible for them to react with bases, including sodium hydroxide and potassium bicarbonate. The products of such reactions are salts of carboxylic acids. The solubility of carboxylic acids in water is negligible, but their salts with alkali metals, due to their ionic structure, dissolve very well in water. These compounds, which are acids in the Bronsted-Lowry theory, dissociate in dilute aqueous solutions. As a result of the transformation, the carboxylate anion RCOO– and the hydronium cation H3O+ are formed.
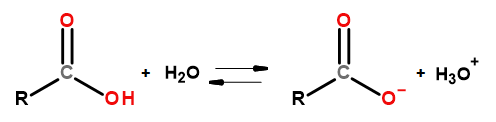
Figure 3Scheme of carboxylic acid dissociation.
Similarly to other acids, it is possible to write out the acid constant (Ka) and its logarithm:
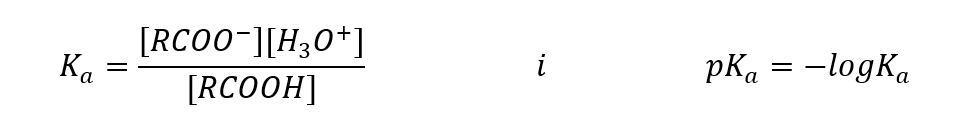
Empirically determined values for carboxylic acids show that they are weakly dissociated compounds, so in practice they are also weak acids. For most of them, the Ka constant takes a value of about 10-5. For acetic acid
Ka = 1.76·10-5, which, after conversion, gives the pKa value equal to 4.75. This equates to the dissociation of only about 0.1% of the molecules, which, compared to strong inorganic acids with a degree of dissociation of 100%, confirms that these are low-strength acids. Compared to alcohols, whose equilibrium constants oscillate in the 10-16 order of magnitude, carboxylic acids are much stronger. Despite their structural similarity, this fact is due to the stability of the carboxylate anion relative to the alkoxide anion. Note that the negative charge in alkoxides is on a single oxygen atom, which is highly electronegative. In contrast, in carboxylate anions, the charge is delocalized between the two oxygens of the functional group. This makes it more stable compared to the anion produced by alcohols.
Obtaining carboxylic acids
- Oxidation of substituted alkylbenzenes with KMnO4 or Na2Cr2O7 with the product in the form of substituted benzoic acids. It is possible to oxidize primary and secondary alkyl groups,
- Oxidation of the C=C bond in alkenes with at least one vinyl hydrogen atom using KMnO4,
- Oxidation of primary alcohols and aldehydes. Alcohols using the Jones reagent, aldehydes using the same reagent and basic silver oxide,
- Hydrolysis of nitriles under the influence of strong, hot solutions of acids or bases,
- The reaction of Grignard compounds with carbon dioxide, with an intermediate product in the form of magnesium carboxylate. The transitional compound then reacts with the acid.
Reactions of carboxylic acids
Due to their structure, carboxylic acids, in addition to their characteristic reactions, also undergo transformations similar to alcohols and ketones. Like alcohols, they can dissociate a proton to form a nucleophilic reagent. Like ketones, they are subject to attacks by other nucleophiles on the carbon atom of the carbonyl group. In addition, their reduction, substitution in the alpha position, nucleophilic substitution of the acyl group and deprotonation (dissociation) are possible.
Reduction of carboxylic acids takes place with LiAlH4, giving the product as a primary alcohol. Sometimes it is necessary to apply heating in a tetrahydrofuran solution to increase efficiency. Another method is a reduction using borane in tetrahydrofuran solution, also obtaining 1o alcohols.