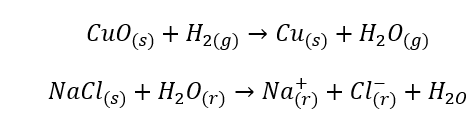Deliberations on various phenomena and processes must be multilayered. We should take into account both the microscopic and macroscopic properties. As the names suggest, this division is made by separating the orders of magnitude of the analysed items. The macroscopic domain, closely linked to the thermodynamics of chemical reactions, allows us to observe and measure physical and chemical properties such as density, temperature or solubility. By microscopic analysis, we can observe the motions and interactions of molecules. We can describe molecules in terms of their speed and kinetic energy. If we are interested in a mixture, we may take into account its homogeneity and, consequently, the type of system it exists in.

Classification of physico-chemical systems
When analysing the homogeneity of material systems, the macroscopic scale classifies them into two principal groups: homogeneous and heterogeneous systems. This categorisation is by their physical characteristics: the physical state, solubility and melting point, which may differ or not. Every system includes all substances that take part in a particular reaction or process. The portion of the system that clearly stands out by maintaining the same properties is referred to as a phase. There are three different types of phases: solid, liquid and gas. The surface located between them and the remainder of the system is called the interface. Depending on the free enthalpy (ΔG) or entropy (ΔS), if ΔS>0 or ΔG<0, the transformation of one phase into another may take place automatically.
Phase equilibrium diagram
A graphic representation of the existence of different phases is called the phase equilibrium diagram. It includes the existence of specific phases (if certain temperature and pressure ranges are reached) and their mutual conversions. A schematic phase equilibrium diagram for a one-component system illustrates the existence of three main phases depending on the process conditions. The lines it shows indicate the states of equilibrium between two respective phases, that is between solid and gas, between solid and liquid, and between gas and liquid. Thus, if we know one of the critical parameters, we can determine the conditions that will cause melting, freezing, evaporation, condensation, sublimation or resublimation of the ingredient in question. Additionally, the diagram includes a triple point where all phase lines connect, which indicates that all the three phases can coexist in specified conditions. The diagram also includes two important points: critical pressure and critical temperature. The critical pressure is indicated by the point representing the maximum pressure at which we can convert a liquid into a gas, or the maximum possible pressure of saturated vapour above the liquid. The concept of critical temperature defines the highest temperature at which a gas can condensate as the pressure increases. Therefore, the critical point existing at critical temperature and critical pressure conditions defines a state in which the liquid and gaseous phases are impossible to differentiate.
Gibbs phase rule
It is a specific relationship which can be applied to any system existing in thermodynamic equilibrium. The equation representing that rule contains the number of phases (f) present in the system with the number of independent constituents (n) and the number of degrees of freedom (s). The s number corresponds to the quantity of adjustable intensive variables which, if changed, will not disturb the number of phases in the equilibrium of the isolated system. Intensive variables are any physical values that in no way depend on the system (e.g., geometric sizes or volume) or on the number of its molecules, its weight, specific heat, etc. The equation is as follows:
The independent components of the system, designated as n, practically represent the lowest number of system components required to build each of its phases in any quantity. If we analyse a system whose components may react with one another, we calculate the n value by subtracting the number of independent reaction equations from the total number of components that form the system. Thus, if we assume the following notation:
![]()
the number of independent equations is 1 – we examine a particular reaction that occurs in the system. An independent equation is any system of chemical equations in which it is impossible to obtain any of them by combining the others. Whereas the number of components for such a system is 3: they are CaCO3, CaO and CO2. This information also allows us to calculate the number of independent components:
n = the number of components – the number of independent equations, and thus:
n = 3 – 1 = 2
We can use the same example to calculate the number of degrees of freedom using the equation for the Gibbs phase rule:
s = n – f + 2 = 2 – 3 + 2 = 1
This value indicates that in order to maintain a stable quantity of phases in system equilibrium, we can operate only one intensive variable.

Heterogeneous systems
Any system that includes more than one phase (so a non-homogeneous system) is also heterogeneous. The type of the phases is irrelevant; a heterogeneous system may at the same time contain several solid and liquid phases but only one gaseous phase. This is due to the fact that gases mix in a perfect way.
An interesting example of a heterogeneous system is granite – it contains many solid phases, including quartz, potassium feldspar, plagioclase and biotite. Many food products can also be called heterogeneous, perfect examples being multi-ingredient muesli or salad sauces that consist of fat, spices and other additives.
An interesting type of non-homogeneous mixtures are colloids (solutions whose constituents can only be distinguished with a microscope). However, in most cases they can be separated with simple methods or tools. For instance, to separate a mixture consisted of water and oil, we can use decantation – the ingredients will be visible to the naked eye. Other methods we can use to effectively separate non-homogeneous mixtures include: filtration, mechanical segregation, or centrifugation. It is similar for a mixture of minerals that differ in properties: quartz, mica and feldspar can be separated even with a hammer.
Homogeneous systems
Unlike heterogeneous systems, a homogeneous system is characterised by the presence of only one phase. In such a case, we cannot distinguish the constituents with the naked eye. Simple methods of separation are also ineffective, and only methods based on physical characteristics may turn out to be useful. An example of such a method is the separation by distillation, which is based on diversified boiling points of a specific system. Other methods effective in separating homogeneous mixtures are the crystallisation and chromatography. A special type of a homogeneous system is a pure substance whose only phase contains only one component. Another example are all types of homogeneous solutions and mixtures – despite the presence of only one phase, the system may include many components. As the phase present in the system can be a liquid, a solid or a gas, solutions may also take such forms. Examples of such systems are liquid mixtures (vinegar, water), gaseous mixtures (atmospheric air) and solid mixtures, including mineral ores and all types of alloys, such as bronze or brass.
Transitions in systems
Two types of transitions may occur in systems, these being phase (sometimes called physical) and chemical transitions. The former ones include all transitions that do not produce new chemical substances. They are also not characterised by the fading of the components already present. However, as the name indicates, they lead to such changes in the structure or the physical state.. It is also possible that a new phase will be formed while the original phase will fade out. Examples of phase transitions include resublimation and polymorphic transitions of elements. Chemical transitions show a completely different mechanism. These are reactions which produce new substances that replace the original constituents. Such transitions may be carried out in two variants, for example during hydrolysis in one or more phases as well as during burning.
Chemical reactions
- Homogeneous reactions occur when the entire system, including substrates and products, is in one phase, for example:
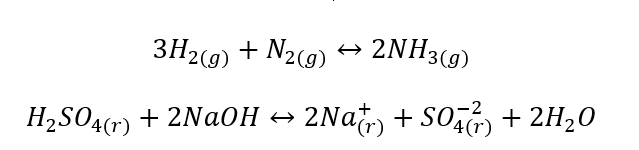
- Heterogeneous reactions are such reactions where the system, including substrates and products, is at least in two different phases. Such reactions always occur at the interface, for example: