It is one of the oldest instrumental methods used in chemical analysis. Its foundations were laid by L. Galvani, C.A. Volta, J.W. Gibbs, and W. Nernst between the 18th and 19th centuries. Measurements performed with potentiometric methods use the electromotive force (EMF) of a cell built of two electrodes immersed in the tested solution. The value of that force directly depends on the potentials of the electrodes used. These in turn depend on the activity of the ions contained in the electrolyte solution and on the nature of electrode processes.
Diagram of electrode processes exemplified by the Daniell cell
The Daniell cell is made of two half-cells, each composed of a metal immersed in the solution of its own salt. The component half-cells are the systems of Cu(s)/Cu2+(aq) and Zn(s)/Zn2+(aq). Each of them undergoes one of the following redox reactions:
- reduction
Cu2+(aq) + 2e– → Cu(s)
- oxidation
Zn(s) → Zn2+(aq) + 2e–
When we combine both reactions, we obtain the full equation for the reaction occurring in the cell:
Zn(s) + Cu2+(aq) + 2e– → Zn2+(aq) + 2e– + Cu(s)
In order to measure a particular electromotive force of the cell, we have to link the half-cells using a salt bridge and connect the electrodes to a voltmeter. Thus we can measure the difference of potentials between individual electrodes, which represents the cell’s EMF value. According to the IUPAC convention, the diagram of the Daniell cell looks as follows:
ϴZn|Zn2+||Cu2+|Cu⊕
The single lines stand for the interface, and the double lines represent the salt bridge. The potential at the interface is a result of the oxidation and reduction reactions. For the cells, we assume that the electrode where oxidation occurs is the anode with the ϴ sign, while the other one is a positive cathode where reduction occurs. The notation begins with the anode, and the cathode is noted on the right side. The electromotive force of such a cell can be calculated based on the following formula:
EMF = cathode potential – anode potential
Standard electrode potential
This term has been introduced for systematisation, and a determined potential of an electrode is considered “standard” if it satisfies a few conditions. The measurement should be performed at 298 K, the activity of active ions must be equal to 1 mol/dm3, and the reference electrode must be a standard hydrogen electrode, as it is assumed that the potential of that electrode is 0.0000 V.
Electromotive force of the cell
The EMF value changes along with the changing activity of the ions present in the cell. The equation which reflects this relationship is called the Nernst equation:
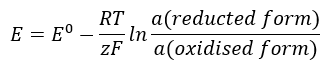
It uses the following symbols: electrode potential (E), standard electrode potential (E0), gas constant (R) equal to 8.312 J/K·mol, temperature (T), the number of electrons involved in the electrochemical reaction of electrons (z), Faraday constant (F) equal to 9.64853·104 C/mol, and the activity of ions in the reduced and oxidised forms. For solids, the activity (a) is assumed to be 1.

Potentiometric instrumentation
The basic set for potentiometric measurements consists of two components: a pair of electrodes immersed in the tested solution and a measuring device. The first component is the measuring cell, while the second enables the actual measurement of its electromotive force. The most important part of a cell are the electrodes. According to their mechanism of action, they can be classified into four groups.
Salt bridge
An important element of any cell is the salt bridge, which allows the reference electrode to contact the tested solution. The fundamental characteristic of a cell with transference is the contact between electrolytes of different concentrations and compositions. To avoid mixing of the bridge’s electrolyte with the tested solution, small contact surfaces are used.
Electrodes of the first kind
These electrodes are made of a metal or gas immersed in the solution containing their own ions. They are also called electrodes reversible to the cation, as on their surface, the following reversible reaction occurs:
M0 ↔ Mn+ + ne
An example of electrodes of the first kind is the hydrogen electrode.
Electrodes of the second kind
Such electrodes are built of a metal covered with its sparingly soluble salt. The system is immersed in a freely soluble salt, which contains the same anion as the sparingly soluble salt. These electrodes are called electrodes reversible to the common anion, as on their surface the following reaction occurs:
M0 + A– ↔ MA + e
Examples of electrodes of the second kind include the silver-silver chloride or calomel electrodes.
Electrodes of the third kind
These are composed of metals surrounded by two layers: the first, thin layer made of a sparingly soluble salt of that metal, and the second layer made of a slightly better soluble salt that contains the same anion. They are electrodes reversible to common cation, as on their surface the following reaction occurs:
M0(a) + M(b)A ↔ M(a)A + M+(b) + e

Redox electrodes
The structure of such electrodes is characterised by the use of a chemically inert metal, for example platinum or gold, which is immersed in a solution containing a substance in both reduced and oxidised forms. An example of such electrodes is the quinhydrone electrode.
Functions of electrodes in a system
In terms of application in an analytical system, we distinguish:
- reference electrodes, which maintain a constant potential regardless of measuring conditions or the concentration of the tested solution.
- indicator electrodes, whose potential changes if the tested solution contains ions, to which they are sensitive.
Reference electrodes
Potentiometric measurement is based on the comparison the potential of the indicator electrode with the reference electrode. For this reason, such electrodes must be characterised by:
- stable potential,
- reproducible potential and temperature independence,
- versatility and ease of use,
- low electrical resistance,
- reproducible and low diffusion potential,
- little flow of the electrode’s electrolyte to the tested solution.
A commonly used reference electrode is the silver-silver chloride electrode (Ag/AgCl/KCl), which reacts to the presence of chloride ions. It is built of a silver wire coated with a layer of silver chloride. Another example is the calomel electrode (Pt/Hg/Hg2Cl2/KCl), whose potential also depends on the activity in a chloride ion solution.
Indicator electrodes
For the electrodes from this category their potential changes in the presence of tested ions in the solution. This means that their potential directly depends on the activity, so also the concentration, of the solution in which they are placed. The most commonly used are electrodes immersed in the solutions of their own ions, such as silver, mercury or hydrogen electrodes. However, ion-selective membrane electrodes are used in modern potentiometry. They have some common characteristics, for example, their active part is the membrane, and the ionic reaction between the ions from the solution and the membrane determines the difference in potential at their interface. Such electrodes can be divided based on their structure into glass, solid membrane, liquid membrane, and double membrane electrodes. The most often used are the glass electrodes, which are built of a glass tube terminated with a thin-walled bulb made of electrode glass. It accommodates the internal, silver-silver chloride electrode immersed in the internal solution with a constant activity of ions.