The packaging of drugs and supplements contain various designations, symbols and letter abbreviations that are not easily comprehensible to consumers. Such designations include the Lot number on the packaging. In our article, we will explain how it should be interpreted and where the requirement to use this abbreviation on packaging and leaflets came from.
Abbreviations on drug packaging – what do they indicate?
Medicinal devices, drugs and supplements constitute a specific group of products. They are subject to strictly defined legal regulations, covering e.g. the scope and type of information provided on the packaging. What do codes and abbreviations on drug labels or leaflets mean? They indicate, among others:
- the abbreviated name of the manufacturing company (e.g.: Polpharma as PPH)
- country of drug origin,
- the content of the medicinal substance in a specific measurement unit,
- drug expiration date,
- batch number,
- rules for using the drug or its reactivity with selected substances,
- information on the drug availability: whether it is available to the general public or on prescription.
Some drugs distributed in Poland may also have special labelling in the form of a QR ID code. In addition, many packaging manufacturers also use colour markings, indicating the range of effects of medicinal substances.
See the range of pharmaceutical raw materials and additives from the PCC Group.
The designation Lot in the light of the Polish law
Obviously, the choice of designations on drug packaging is not random. Pharmaceutical companies are obliged to comply with current guidelines on product labelling. Guidance on the use of Lot and other designations is included in, e.g.:
- Notice of the Minister of Health of 29.09.2020 on the announcement of the consolidated text of the Regulation of the Minister of Health on requirements for labelling medicinal product packaging and the content of the leaflet,
- Pharmaceutical Law of 6.09.2001,
- Directive 2001/83/EC of the European Parliament and of the Council of 6 November 2001 on the Community code relating to medicinal products for human use.
The above-mentioned acts of Polish and EU law refer to the issue of designation used on product packaging and leaflets. One of such designations is Lot – what does it mean? Let’s check.
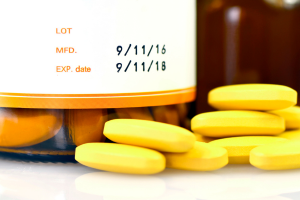
Lot: what does it mean when stated on the package?
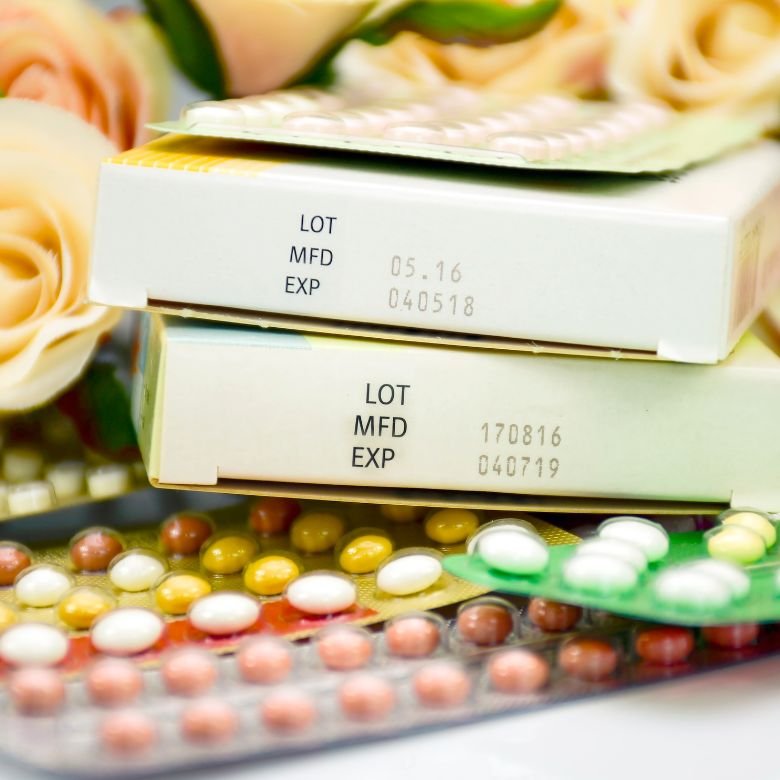
The abbreviation: Lot is a common symbol that appears as standard on the sides of the packaging of various drugs and in the content of leaflets. How should it be understood? The Lot indicates the drug batch number. According to the Pharmaceutical Law, a batch should be understood as:
- a specific amount of a medicinal product or pharmaceutical raw material or packaging material, produced in a process consisting of one or more operations in such a way that it can be considered homogeneous (Art. 1 section 37 of the Pharmaceutical Law).
In accordance with EU regulations, the production of each batch of drugs is subject to specific guidelines – assigning a number to a particular batch means that it has passed the necessary checks and has been approved for marketing.
Optionally, Lot may also be replaced by the abbreviation “S”, provided that the expiry date is marked as Tw (meaning EXP). On many packages of medicines, cosmetics and dietary supplements, you may also see that the abbreviation Lot is accompanied by a standard description, i.e.: batch number. In some countries outside the European Union, the Lot number is known as: batch number. This information must be included on the carton, container or leaflet.
Lot vs EXP – what’s the difference between the two abbreviations?
As indicated by the legislator, the Lot as the designation of the product batch number should appear next to the designation EXP (in capital letters). What does EXP stand for? On the packaging of drugs, it indicates the product expiration date. As a standard, next to the EXP symbol, there is information about the expiration year and month. According to Polish law, the drug expiration date should be presented in the following way:
- understandable,
- unencrypted,
- specifying the month and year – the month should be presented as 2 digits or at least 3 letters, and the year – as 4 digits.
Thanks to the legible and understandable EXP data, consumers, pharmacists and controllers can easily check whether a given product can be freely used, whether it is fresh and whether its properties are preserved.
Why were designations such as Lot introduced?
The Polish pharmaceutical industry continues to develop, and new drugs and supplements are still launched on the market. Thanks to the unified labelling system using Lot, EXP, etc., consumers and controllers (including GIF) are able to easily find important information on the packaging. What are the benefits of Lot designations and why are they used? They allow one to, e.g.:
- quickly identify a selected batch (e.g. for market recall),
- examine the genuineness of products manufactured in Poland,
- limit the import of illegal drugs from outside the European Union.
The designation Lot also facilitates the capture and monitoring of batches with a potential adverse effect.
For obvious reasons, unified designations such as Lot are not the only security feature for medicinal products. In addition, 2D codes and ATD identifiers appear more and more often on the packaging, preventing damage to the packaging. Thanks to identifiers and serialisation, drug counterfeiting becomes increasingly difficult, which benefits the safety of consumers.
- Drug serialisation highlights, article in polish, access: https://www.nia.org.pl/serializacja/
- Serialisation of medicinal products) article in polish, access:https://www.gov.pl/web/gif/serializacja-produktow-leczniczych
- Notice of the Minister of Health of 29.09.2020 on the announcement of the consolidated text of the Regulation of the Minister of Health on requirements for labelling medicinal product packaging and the content of the leaflet, article in polish, access: https://isap.sejm.gov.pl/isap.nsf/DocDetails.xsp?id=WDU20200001847
- Pharmaceutical Law of 6.09.2001, article in polish, access: https://isap.sejm.gov.pl/isap.nsf/DocDetails.xsp?id=wdu20011261381